Lead Chloride Manufacturer & Supplier | Lead Chloride For Sale
- Home
- Lead Chloride Manufacturer & Supplier | Lead Chloride For Sale
- Company Name
- Location
- : BÖRÜ INC.
- : Türkiye
If you need help and get more about our biotechnology products, please contact with our team.
What is Lead Chloride?
In this article, we have dealt specifically with the formula for lead chloride as a structural and chemical representation. It is an inorganic compound with lead as the central atom and two chlorine atoms attached to it. Lead chloride is a white solid compound under ambient conditions but is otherwise a crystalline, colorless and odorless compound and is also the most important lead-based reagent. Lead chloride is also expressed formulaically as PbCl2. Cotunnite is a natural mineral form of lead chloride and is therefore a naturally occurring inorganic compound found in mineral ore form. It is insoluble in water, but readily soluble in solutions containing chloride ions. Lead chloride is more stable than lead tetrachloride because lead belongs to group 4, where all elements belonging to group 4 show two oxidation states, +4 and +2. However, as you go down in the group, the stability for the +4 oxidation state decreases and the +2 oxidation states increase. So, unlike CCl4 or SiCl4, PbCl4 is much less stable in nature and readily decomposes at room temperature to form a very stable PbCl2.
If you are looking for a lead chloride manfuacturer and supplier and also lead chloride for sale, do not hesitate to contact us.
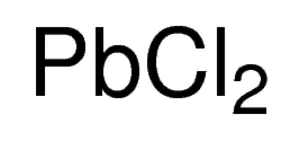
Lead Chloride Structure
The chemical formula for lead chloride is PbCl2, commonly known as lead chloride, but the IUPAC name also stands for lead chloride. The chlorine atoms are bonded to the central atom lead via covalent bonding with a bond angle of 98o, and the isotopic bond distance of each of the chlorine atoms to the central lead atom is 2.44 Å. Now the electronic configuration of the molecules and their atoms needs to be taken into account to explain the nature of the bond. Now the atomic number of the lead atom is 82, which means that its electronic configuration would be Pb 6s2 4f14 5d10 6p2. Thus the outer shell of lead has two electrons to donate or share. On the other hand, chlorine has an atomic number of 17, meaning that it has an electronic configuration of 1s2 2s2 2p6 3s2 3p5, meaning that it has an unpaired electron in the 3p orbital, which makes the shell unstable and needs an electron to complete its octane. So the two chlorine atoms share each of the two pairs of electrons of lead in the 6s shell to complete the octane and hence become a stable molecule. The molecule lead chloride forms a twisted structure.

Lead Chloride Manufacturer
Why choose us for Lead Chloride?
- Biotechnology Laboratory
- Experience
- High Quality
- Right on Time
About Lead Chloride
In each of the molecules present in lead chloride, each of the lead atoms is surrounded by nine chloride ions, like the molecular geometry of a triangular prism molecule with three heads, where nine atoms or groups of atoms or ligands are arranged in this way around the center atom. In solid lead chloride molecules, there are six chloride ions at the corners of the triangular prism with lead as the center atom and three chloride ions beyond the faces of each rectangular prism face. Therefore, the chloride ions are not equidistant from the central lead atom. Seven of the nine chloride atoms are at 280 -309 pm and the other two chloride atoms are at 270 pm.

