Lead Nitrate Manufacturer & Supplier | Lead Nitrate For Sale
- Home
- Lead Nitrate Manufacturer & Supplier | Lead Nitrate For Sale
- Company Name
- Location
- : BÖRÜ INC.
- : Türkiye
If you need help and get more about our biotechnology products, please contact with our team.
What is Lead Nitrate?
Lead nitrate is an inorganic compound. Its chemical formula is known as Pb(NO3)2. It often appears as a colorless crystal or white powder and, unlike many other lead salts, is soluble in water.
The production of this compound was on a much smaller scale than other lead compounds. Historically, it first established itself as a commercial material in America. Also later, its main use was as a raw material in the production of pigments for lead paints, but such paints were replaced by less toxic paints based on titanium dioxide. Other industrial uses include heat stabilization in nylon and polyesters and in photothermographic paper coatings. Around 2000, lead nitrate began to be used in gold cyanidation.
Lead nitrate is toxic, an oxidizing agent and is categorized by the International Agency for Research on Cancer as possibly carcinogenic to humans. As a result, it should be handled and stored with appropriate safety precautions to prevent inhalation, ingestion and skin contact. It is used under supervision as its use may pose a hazard.
If you are looking for a lead nitrate manfuacturer and supplier and also lead nitrate for sale, do not hesitate to contact us.
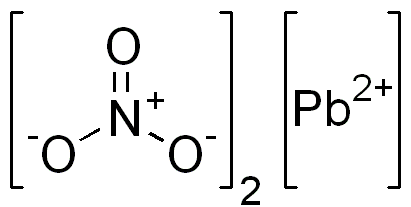
Use of Lead Nitrate
Due to the hazardous nature of lead nitrate, alternatives are preferred for industrial applications. The formerly major application of lead paint has been largely replaced by titanium dioxide. Other historical applications of lead nitrate, such as matches and fireworks, have also declined or ceased. Current applications of lead nitrate include its use as a heat stabilizer in nylons and polyesters, as a coating for photothermographic paper, and in rodenticides.
On a laboratory scale, lead nitrate provides one of two convenient and reliable sources of dinitrogen tetroxide. By carefully drying lead nitrate and then heating it in a steel vessel, nitrogen dioxide is produced along with dioxygen following the decripitation equation shown above. Alternatively, nitrogen dioxide is formed when concentrated nitric acid is added to copper chips, in which case significant amounts of nitrogen monoxide can also be produced.

Lead Nitrate Manufacturer
Why choose us for Lead Nitrate?
- Biotechnology Laboratory
- Experience
- High Quality
- Right on Time
Lead Nitrate History
Since the Middle Ages, lead nitrate has been produced as a raw material, aiming at the production of colored pigments in chrome yellow, chrome orange (lead(II) hydroxide chromate) and lead paints. These pigments were used to dye and print calico and other textiles.
When the calendars showed 1597, this compound was identified for the first time. It was so named because of its sweet taste. Later, although not originally understood, the aging properties of lead nitrate led to its use in matches and special explosives such as lead azide.
The production process followed a chemically simple route that effectively dissolved the lead in aqua fortis (nitric acid) and then collected the precipitate, and this has continued to the present day. However, production remained small-scale for centuries and commercial production of lead nitrate as a raw material for the production of other lead compounds was not reported until 1835. In 1974, US consumption of lead compounds, excluding pigments and gasoline additives, was 642 tons.

