Barium Nitrate Manufacturer & Supplier | Barium Nitrate For Sale
- Biotechnology Company
- Barium Nitrate Manufacturer & Supplier | Barium Nitrate For Sale
- Company Name
- Location
- : BÖRÜ INC.
- : Türkiye
If you need help and get more about our biotechnology products, please contact with our team.
What is Barium Nitrate?
Barium nitrate is an inorganic compound that is officially known by many other names as well. Some prefer to call it barium dinitrate, while others may know the compound as Nitrobarite. However, unofficially, people may refer to this particular compound as barium salt.
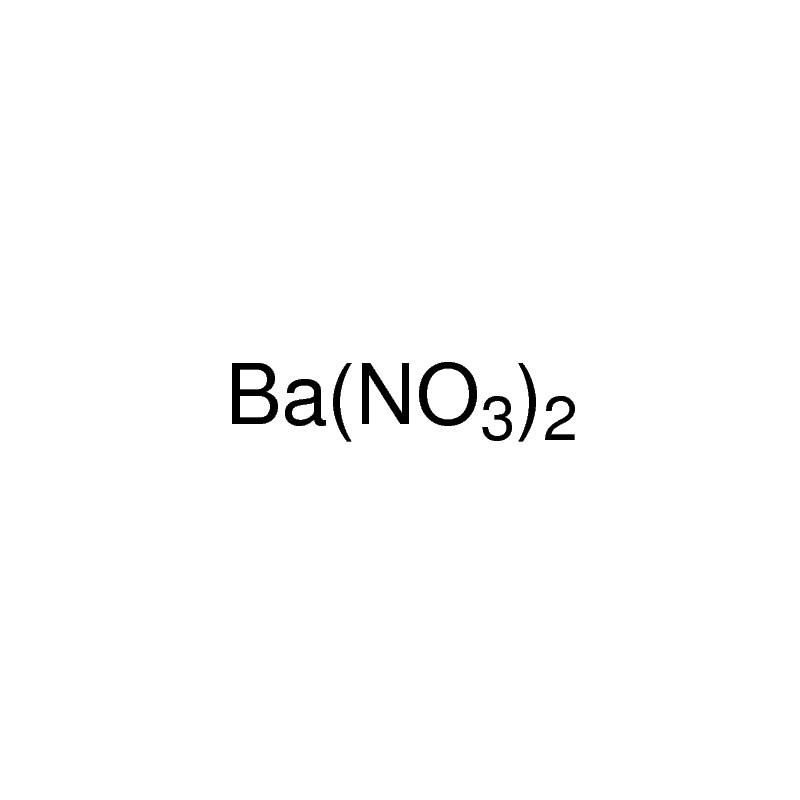
Barium nitrate formula is Ba(NO3)2. If you are appearing for your final year examinations soon, you need to have distinct knowledge about this particular chemical, along with all of its properties. By the end of this chapter, you will also learn about this salt’s various applications. Furthermore, major preparation processes are also discussed in detail.
The key to understanding how any compound functions is to learn about its various properties. Refer to the table below to gain insight into its various basic features and natural properties.
Barium Nitrate Structure
Barium nitrate is used in the production of barium oxide containing materials.
Used in green signal lights.
Used in making ceramic glazes.
Used as a rodenticide.
Used in detonators.
Used in primers and tracer bullets.
Used in the making of paints.
Used as an oxidizing agent.
Used in making explosives.
Used as a propellent and blowing agent.

Barium Nitrate Manufacturer
Why choose us for barium nitrate?
- Biotechnology Laboratory
- Experience
- High Quality
- Right on Time
Frequently Asked Questions
What are the Uses of Barium Nitrate?
Barium nitrate is used in the manufacture of substances containing barium oxide. This compound is also used in the barium oxide production process, in the vacuum tube industry and in pyrotechnics for green fire. Barium nitrate has also been a key ingredient in the explosive charge “SR 365”.
What Happens When Barium Nitrate is Heated?
When burned, barium nitrate emits a green flame. It decomposes to barium oxide, oxygen, and nitrogen dioxide when heated. This converts barium nitrite into nitric oxide. To produce barium sulfate it combines with either sulfate or sulfuric acid.

