Lead Picrate Manufacturer & Supplier | Lead Picrate For Sale
- Home
- Lead Picrate Manufacturer & Supplier | Lead Picrate For Sale
- Company Name
- Location
- : BÖRÜ INC.
- : Türkiye
If you need help and get more about our biotechnology products, please contact with our team.
What is Lead Picrate?
Lead picrate is an organic picrate salt. It exists in the form of a hydrate, characteristic of its structure. Lead picrate is dangerous. Because it is a sensitive compound that can explode at any time. If lead picrate is found in dry form, this is a great danger. It is also a very dangerous inorganic on its own. It is already known to be used especially in weapons production. It has often been reported that lead picrate was the most widely used compound in wartime Germany.
If you are looking for a lead picrate manfuacturer and supplier and also lead picrate for sale, do not hesitate to contact us.
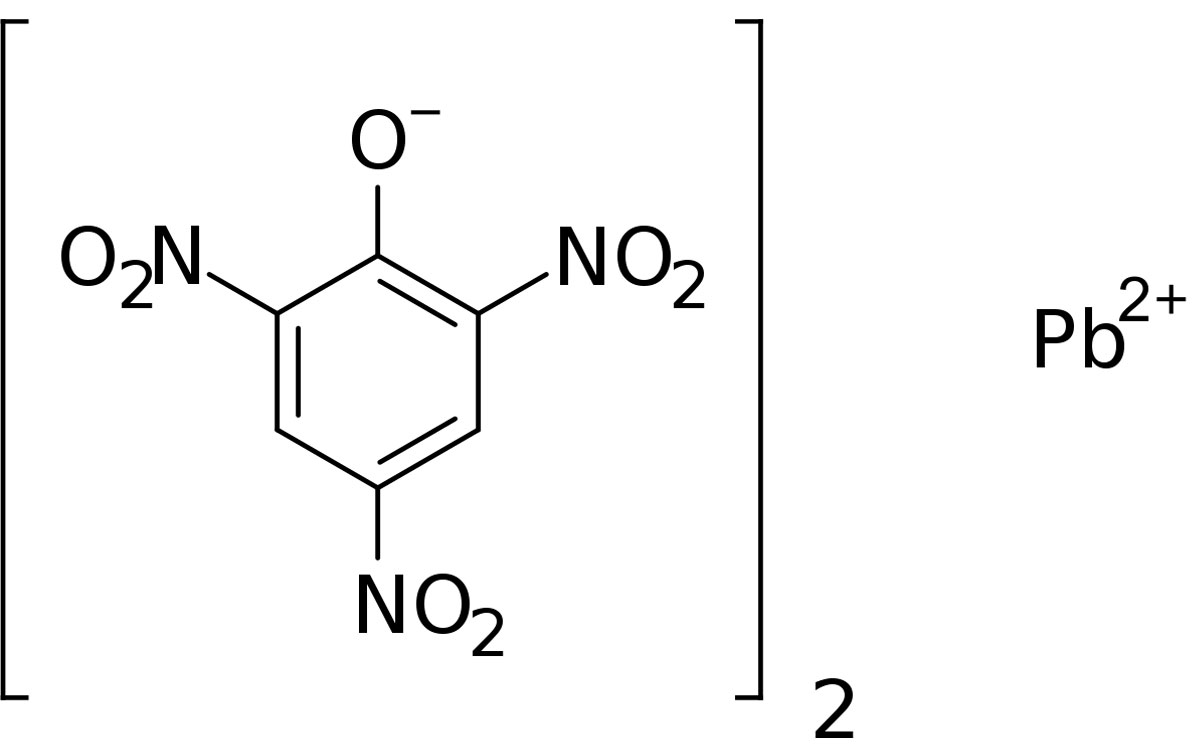
About Lead Picrate
Picric acid is known to react with metals to form highly unstable metallic picrates, which have been the cause of a number of serious explosion accidents. In this paper, lead picrates were synthesized by various methods and their explosive properties such as thermodynamics and sensitivity were presented. Differential scanning calorimetry (DSC) results revealed that lead picrates have several heat decomposition patterns depending on the synthesis method and have a lower exothermic reaction initiation temperature than sodium picrate. The heat of decomposition of lead picrates was found to be lower than that of picric acid. Lead picrate from lead acetate and picric acid had lower activation energy of thermal decomposition than other lead picrates. Thermogravimetry analysis and Karl Fischer analysis confirmed that lead picrates contained crystalline H2O that dehydrated above 375 K. Lead picrates had no fusion properties. Drop hammer test results showed that lead picrates had high impact sensitivity, while lead picrates had low friction sensitivity in friction tests attributable to the presence of crystalline H2O. The ignition temperatures of lead picrates were found to be in the range.
Lead picrate was noted to be particularly sensitive. Lead picrate has also been specifically investigated by a number of researchers. In particular, they have been classified as a primary group explosive due to their potentially explosive nature. In Germany, it is often used as a blasting cap. In addition, we can say that lead picrate was at its peak a few decades ago. This is because fewer studies of metallic picrates (including lead picrate) have been carried out recently using modern analytical techniques. Therefore, until recently, the chemical and pyrotechnic properties of picrates were still unclear.

Lead Picrate Manufacturer
Why choose us for Lead Picrate?
- Biotechnology Laboratory
- Experience
- High Quality
- Right on Time
How is Lead Picrate Produced?
When producing lead picrate, lead oxide is added to picric acid. In addition, there is a different method. In this method, a solution of picrite acid is added to a solution of sodium picrate, which is already a base. Thus, we obtain a hydroxide solution. The resulting solution is then slowly added, with constant stirring, to a solution of lead nitrate close to boiling point. The next step is to allow the mixture to cool to room temperature. The resulting basic lead picrate is too concentrated and can quickly reach the bottom, which is normal. The mixture is then filtered and washed with distilled water. It is not recommended to make too much of this salt at any one time, nor is it recommended to store it dry due to its explosive nature. The production, storage and transportation of this compound may require an explosives permit depending on the country.

