Potassium Nitrate Manufacturer & Supplier | Potassium Nitrate For Sale
- Home
- Potassium Nitrate Manufacturer & Supplier | Potassium Nitrate For Sale
- Company Name
- Location
- : BÖRÜ INC.
- : Türkiye
If you need help and get more about our biotechnology products, please contact with our team.
What is Potassium Nitrate?
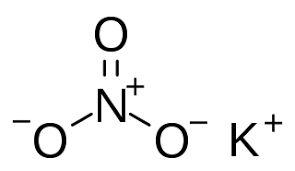
Potassium nitrate, also called saltpeter or nitre, has a white color. It is symbolized as KNO3. It is essentially an ionic salt. Some of the many uses of potassium nitrate include the production of fertilizers, pesticides, glass, fireworks, explosives and rocket fuel. It is also used as a food preservative and when added to meat, it reacts between myoglobin and hemoglobin in the blood, making the meat appear red. It is also sometimes used in toothpaste. Potassium nitrate is dangerous to humans at high levels, so its use is carefully controlled when it comes to human consumption.
It is often called saltpeter (also called nitre). It is found in nature in impure form and its name derives from a Latin word meaning “rock salt”, as it is often found as a white material deposited on the surface of rocks. Saltpeter can occur on the soil surface in various warm climates, including Egypt, Spain and Iran. In such places, feces, urine and decaying plants react with moisture and alkaline soil to form nitrates. These nitrates dissolve in rainwater and when this water evaporates, white deposits of potassium nitrate are left behind. Many caves around the world have large deposits of saltpeter due to the large amounts of bat dung and urine found there.
If you are looking for a potassium nitrate manfuacturer and supplier and also potassium nitrate for sale, do not hesitate to contact us.
History and Production
The invention of gunpowder in China changed everything. When combined, the explosive effect of potassium nitrate, sulphur and charcoal rewrote the rules of the game. After this invention, when China went to war, they used this gunpowder mixture to fight their enemies. They then redirected this power to a different alternative and improved their weapons.
Historically in the United States, in the early to mid-19th century, there was intensive mining of potassium nitrate in caves in the South. As the need for gunpowder grew, so did the need for potassium nitrate. This, in turn, led to increased demand. shifted its production from caves, where only a limited amount of the chemical could be produced, to industrial laboratories with much higher production capabilities. The most common industrial production method uses potassium nitrate in a double displacement reaction with nitric acid.
- Modern industrial production
More than 200,000 tons of potassium nitrate are produced annually in the United States, almost 90 percent of which is used as fertilizer. The 10 percent not covered by this production finds its way into other industries such as the manufacture of matches, fireworks, glass and ceramics. The annual worldwide production of potassium nitrate is close to 40 million tons. Russia has the largest share in this production.

Potassium Nitrate Manufacturer
Why choose us for Potassium Nitrate?
- Biotechnology Laboratory
- Experience
- High Quality
- Right on Time
Chemical Properties
Although not itself flammable, potassium nitrate accelerates the combustion of flammable materials. Potassium nitrate is a strong oxidizer and, when heated, decomposes into potassium nitrite and oxygen, which aids the combustion process in explosives. This is why potassium nitrate is called an oxidizing agent. It has many uses, including the production of fertilizers, pharmaceuticals, gunpowder, fireworks and explosives.

