Silver Nitrate Manufacturer & Supplier | Silver Nitrate For Sale
- Home
- Silver Nitrate Manufacturer & Supplier | Silver Nitrate For Sale
- Company Name
- Location
- : BÖRÜ INC.
- : Türkiye
If you need help and get more about our biotechnology products, please contact with our team.
What is Silver Nitrate?
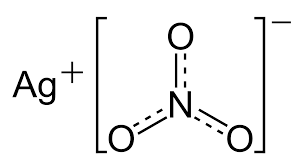
Silver nitrate is a chemical compound. AgNO3 is its symbolic notation. Due to the ionic nature of this compound, it is easily soluble in water and dissociates into its constituent ions. Silver nitrate is the precursor of many silver compounds, including silver compounds used in photography. AgNO3 is highly stable when exposed to light, compared to the silver halides used in photography due to their sensitivity to light.
If you are looking for a silver nitrate manfuacturer and supplier and also silver nitrate for sale, do not hesitate to contact us.
Use of Silver Nitrate
Silver nitrate has a wide range of uses. There are many fields it addresses. Some of these are biology, chemical synthesis and medicine. Some of these uses are listed below.
Silver nitrate is a versatile compound because the nitrate ion can be replaced with other ligands that can bind to the silver ion. Due to this compound’s ability to form a silver halide precipitate when treated with halide ions, it is used in making photographic films. Many silver-based explosives can be prepared by the precipitation reaction of silver nitrate. For the extraction of halides, the silver nitrate compound is often consulted. The branch of chemistry known as analytical chemistry uses this reaction to check for the presence of halide anions such as iodide, bromide or chloride ions. Since the silver cation binds inversely with alkenes, alkene mixtures can be separated with the help of this compound. When diluted with water to a concentration of 0.5%, silver nitrate can act as an antiseptic in many medical installations. A diluted solution of AgNO3 can be applied to the eyes of a baby born to a mother with gonorrhea, which fights gonorrhea bacteria and protects the baby from the onset of blindness. This compound is also known to be used in the treatment and removal of unwanted warts in humans.

Silver Nitrate Manufacturer
Why choose us for Silver Nitrate?
- Biotechnology Laboratory
- Experience
- High Quality
- Right on Time
About Silver Nitrate
What are the uses of silver nitrate?
Examples of uses of silver nitrate include its use for deprotection and oxidation reactions. The Ag+ ion reversibly binds alkenes and silver nitrate, which selectively adsorbs it, can be used to isolate alkene mixtures. The resulting adduct can be decomposed with ammonia (to release the free alkene). It has previously been used for dyeing purposes. This compound is also used in medicine because of its antiseptic properties.
Is silver nitrate dangerous?
At the end of the day, silver nitrate is an oxidant. Despite its widespread use (especially in extremely low amounts) for preventing gonorrhea and stopping nosebleeds, silver nitrate is often highly toxic and corrosive. In addition, prolonged exposure to silver nitrate does not cause any immediate side effects other than the formation of purple, brown or black stains on the part of the skin in contact with silver nitrate. However, prolonged exposure to this compound often causes damage to the eyes.
How is silver nitrate prepared?
Nitric acid is used to prepare silver nitrate. Silver ingots and silver foils are among the preferred silver objects used in this combination. The resulting products include silver nitrate, water and nitrogen oxides. It is important to note that this reaction must be carried out under a fume hood, as toxic oxides of nitrogen are produced during the chemical reaction.

